Pune: The Serum Institute of India (SII) will begin production of Russia’s Sputnik V in September and it is intended to produce over 300 million doses of the vaccine in India per year, said Kirill Dmitriev, CEO of the Russian Direct Investment Fund (RDIF, Russia’s sovereign wealth fund). Earlier in the day, RDIF and SII also announced their cooperation to produce the Russian Sputnik vaccine against coronavirus.Also Read – US Says it’s Ready to Ship Pfizer, Moderna Vaccines, But Waiting for India’s Green Light
Global Covid-19 caseload tops 185.9 mn; UK reports 35K cases in 24 hrs
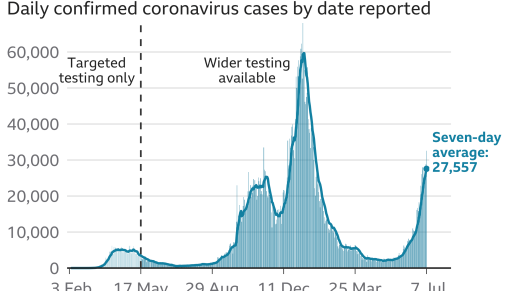
The overall global Covid-19 caseload has topped 185.9 million, while the deaths have surged to more than 4 million and vaccinations soared to over , according to the Johns Hopkins University.
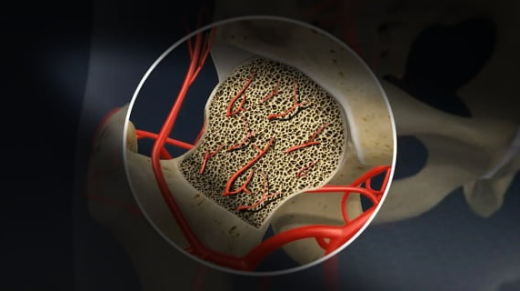
Bone Death, New Post Covid 19 Complication: Symptoms, Causes And Treatment Explained
What is Avascular Necrosis or Bone Death: While our country has just started recovering from the deadly second wave of coronavirus pandemic, preparations are already in process for the expected third wave. Two months after the country started reporting the cases of Mucormycosis (Black Fungus) which is a severe post Covid-19 complication, Doctors now fear that Avascular Necrosis also known as ‘Bone Death’ could be the next possible devastating post Covid-19 condition. At least 3 cases of AVN have been recorded in coronavirus survivors in Mumbai. They reportedly developed necrosis between 45 days to 58 days after COVID-19 treatment.Also Read – What is Bone Death And Why More COVID Patients Are Succumbing to it – All You Need to Know
COVID-19 Lambda strain in India? Here’s what the Centre has to say
New Delhi: The Centre on Friday (July 9, 2021) claimed that Indian SARS-CoV-2 Genomics Consortium (INSACOG) is closely monitoring the Lambda COVID-19 variant, affirming that no case of the Lambda variant of SARS-CoV-2 has been found in India so far.
Statistics of Covid-19 Second Wave
July 12, 2021 Share : Facebook Linkedin Twitter Pinterest
Zydus’ vaccine for kids above 12 to come up for approval this week
New Delhi: The drug regulator will consider emergency use approval for Covid vaccines developed by Zydus Cadila for children above 12 this week. The regulator’s subject expert committee (SEC) will examine data submitted by Cadila, reports Sushmi Dey.
Delhi wins, Kerala loses corona virus battle
The second wave of coronavirus in India has started waning, and in the last 24 hours, the country witnessed 41,506 fresh cases and 895 deaths. However, test results coming out from Kerala is still alarming, and it strongly indicates that the state is choking with the rising number of fresh infections and mounting deaths. In the last 24 hours, the state reported 12,220 fresh coronavirus cases and 97 Covid-related deaths.
Virus antibodies are no match for Delta variant, study shows
People who have previously caught COVID-19 are now more likely to be reinfected thanks to the Delta variant, a study has found.
COVID-19 Lambda strain in India? Here’s what the Centre has to say
The Centre claimed that India is closely monitoring the Lambda COVID-19 variant, affirming that no case of the has been found in India so far.