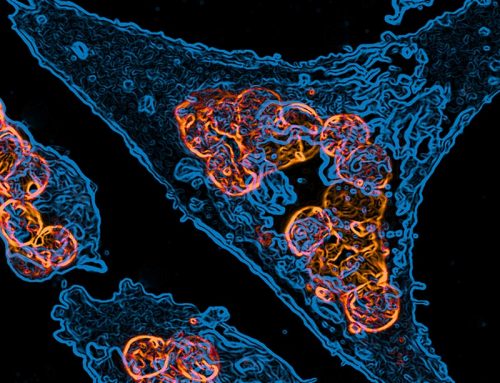
OTTAWA: Researchers probing the gut have for the first time discovered specific factors in its
workings that in the future may help improve treatment for patients facing
the gastrointestinal disease.
The findings of the study were published in the journal ‘Microbiome’.
The findings from Snyder Institute for Chronic Diseases researchers immediately improve the
understanding of factors that help regulate the enteric nervous system, the system of nerves
that control the gastrointestinal tract.
Researchers can now explore novel ways to treat gastrointestinal disorders using
approaches based on these new findings, though the transition to treatment is likely years
away.
The study’s findings may impact future treatments for gastrointestinal diseases and disorders
such as irritable bowel syndrome, inflammatory bowel disease and slow transit constipation,
among others.
“We have uncovered microbial factors that help regulate the function and structural integrity
of the enteric nervous system,” said Dr Keith Sharkey, PhD, a professor in the Cumming
School of Medicine’s Department of Physiology and Pharmacology.
He is also a member of the Snyder Institute and the Hotchkiss Brain Institute. Sharkey is a
senior author of the study and made the discovery with a research team from UCalgary and
the University of Colorado.
“These and other gastrointestinal diseases with manifested changes in enteric neural control
are all hard to treat. Our findings could impact approaches to their treatment,” said Sharkey.
“Be aware, though, that there is a big jump from mice to men. In other words, translating our
findings will be important, and our results are only at the starting point of this journey,” added
Sharkey.
The study examined the effects of microbiome depletion and restoration in animal models
using approaches that cause structural and functional changes in the gut. The researchers
discovered that while microbiome depletion caused a loss of neurons, natural microbiome
recovery restored gut function and promoted the growth of new neurons.
“The findings from our work provide clues as to the mechanisms that control ‘plasticity’ or the
ability of the gut nervous system to be repaired if it undergoes damage,” said Sharkey.
Dr Fernando Vicentini, PhD, first author of the study, said, “The most challenging aspect of
researching the unknown physiological roles of the intestinal microbiota lies in identifying
specific microbial-derived molecules that may affect the host, as there is a myriad of
molecules with the potential to do so.”
He added that after many discussions and brainstorming sessions with the research team,
they were able to hypothesize and demonstrate a link for two different bacterial molecules
involved in the regulation of neuronal integrity.
For Dr Simon Hirota, PhD, the study’s co-senior author, in addition to the thrill of discovery,
this research was exciting in that it allowed him to collaborate with a great team of people
who contributed the complementary expertise needed to test these novel hypotheses.
The research was funded in part by the Canadian Institutes of Health Research, Canada’s
federal funding agency for health research.
The next steps for Sharkey and the research team include exploring other potential microbial
factors and their role in the neural control of the gut.
“There is a rising awareness among the public through discussions in social media, on the
internet and even through the promotion of products such as yoghurts that contain probiotics,
that the gut microbiome plays a vital role in health as well as disease. These conversations
are making their way into physicians’ offices,” said Sharkey.
“As our understanding increases regarding the role the gut microbiome plays in gut health, I
think we will ultimately have new ways to diagnose and treat gastrointestinal diseases on an
individual level,” added Sharkey.